Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...

Nakajima, Kunihisa
no journal, ,
Results on the thermodynamic study of gaseous CsBO will be presented as a keynote speech of the 64th annual conference on Mass Spectrometry. Gaseous cesium metaborate, CsBO
will be presented as a keynote speech of the 64th annual conference on Mass Spectrometry. Gaseous cesium metaborate, CsBO (g), is predicted to be the main cesium vapor species in severe light water reactor accidents according to the thermodynamic analysis, which treats chemical reactions with B
(g), is predicted to be the main cesium vapor species in severe light water reactor accidents according to the thermodynamic analysis, which treats chemical reactions with B C. But quality of the thermodynamic data of CsBO
C. But quality of the thermodynamic data of CsBO (g) used in this thermodynamic analysis is regarded as poor. Thus, Knudsen-effusion mass-spectrometric measurement of CsBO
(g) used in this thermodynamic analysis is regarded as poor. Thus, Knudsen-effusion mass-spectrometric measurement of CsBO was performed to obtain reliable thermodynamic quantities of CsBO
was performed to obtain reliable thermodynamic quantities of CsBO (g). The standard enthalpy values of formation of CsBO
(g). The standard enthalpy values of formation of CsBO (g),
(g), 

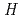
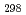
 (CsBO
(CsBO ,g), were obtained by the second and third law treatments and found to be -700.7
,g), were obtained by the second and third law treatments and found to be -700.7 10.7kJ/mol and -697.0
10.7kJ/mol and -697.0 10.6kJ/mol. These two values agree within the error limits and the evaluated thermodynamic data of CsBO
10.6kJ/mol. These two values agree within the error limits and the evaluated thermodynamic data of CsBO (g) are considered to be reliable.
(g) are considered to be reliable.